Your patients will be ready to avoid hypos with the the world's smallest CGM sensor9, that sends glucose readings to their smartphone◊◊ every single minute.


In the example comparison below, the patient's glucose is dropping at 0.2 mmol/L per minute and their low glucose alarm is set at 3.9 mmol/L.


Learn about glucose alarms on the FreeStyle Libre 3 App page.
The FreeStyle Libre 3 app◊◊ allows patients to interpret their glucose data from the FreeStyle Libre 3 sensor on their smartphone anytime‡, anywhere.₸
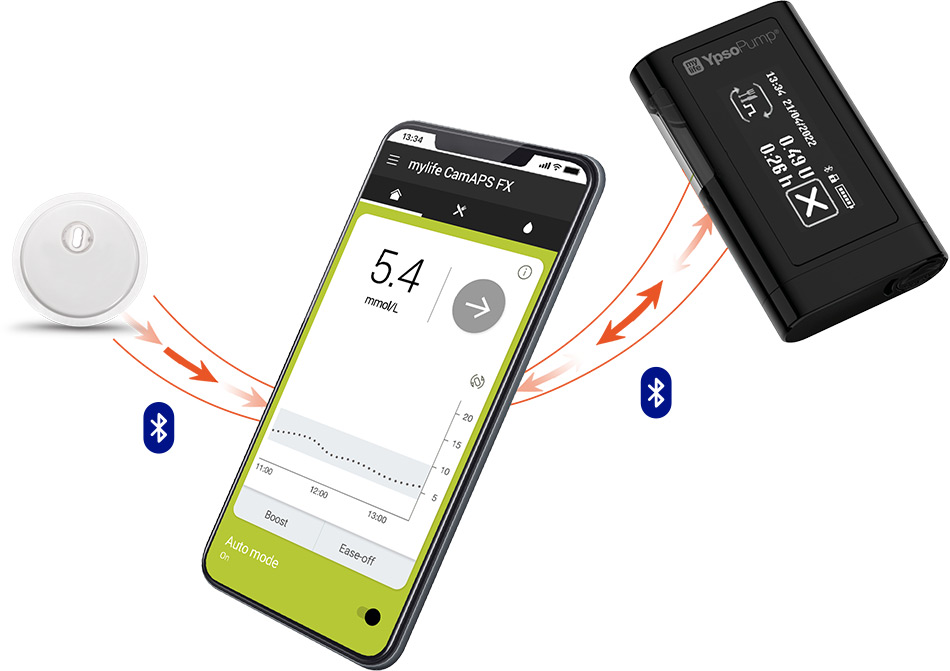
LibreView,₼ our secure14 cloud-based system, allows patients to share their glucose data with Healthcare Professionals. When connected, you will be able to access more in-depth reports to support you in making treatment decisions for your patients.
Patients who use the FreeStyle Libre 3 app◊◊ can remotely share their glucose readings with their caregivers and loved ones through the LibreLinkUp app.§


References & Disclaimers
Images are for illustrative purposes only. Not real patient or data.
1. CGM reliability defined by signal connection. Data on file, Abbott Diabetes Care, Inc.
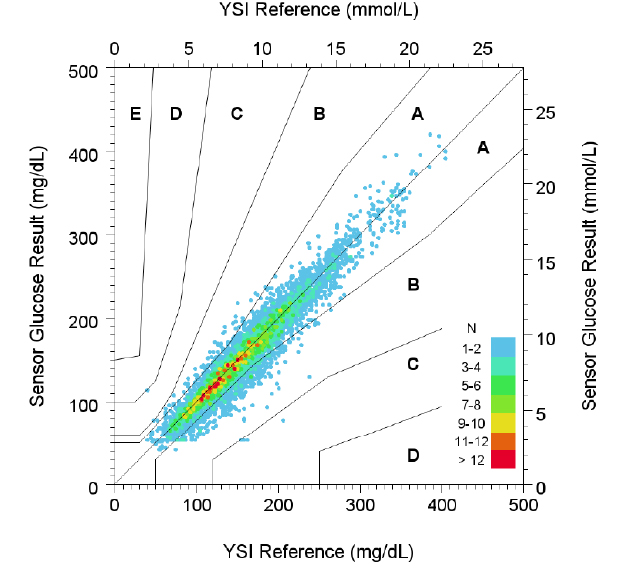
2. FreeStyle Libre 3 User Manual. FreeStyle Libre 3 demonstrates 7.8% overall MARD with 7.5% adult MARD and 8.6% paediatric MARD (age 6-17) in accuracy to YSI.
3. Data on file, Abbott Diabetes Care, Inc. Data based on publicly available information.
4. Dexcom G6 CGM User Guide and Medtronic. Guardian Connect System User Guide.
5. Evans, M. Diabetes Ther (2020): https://doi.org/10.1007/s13300-019-00720-0.
6. Data on file, Abbott Diabetes Care, Inc.
7. Bolinder, J. Lancet (2016): https://doi.org/10.1016/S0140-6736(16)31535-5.
8. Fokkert, M. BMJ Open Diab Res Care (2019): https://doi.org/10.1136/ bmjdrc-2019-000809.
9. Among patient-applied sensors.
10. NICE guideline NG17 (2015) Last updated: 17 August 2022; available at https://www.nice.org.uk/guidance/ng17. Accessed Jan 2023.
11. Real-time continuous glucose monitoring.
12. Multiple daily insulin injections.
13. NICE guideline NG28 (2015) Last updated: 29 June 2022; available at https://www.nice.org.uk/guidance/ng28. Accessed Jan 2023.
14. LibreView is ISO27001/27018/27701 certified and HITRUST CSF Certified.
◊◊ The FreeStyle Libre 3 app is only compatible with certain mobile devices and operating systems. Please check our website for more information about device compatibility before using the app. Sharing of glucose data requires registration with LibreView.
^ Notifications will only be received when alarms are turned on and the sensor is within 33 feet (10 meters) unobstructed of the reading device.
† Finger pricks are required if glucose readings and alarms do not match symptoms or expectations.
₼ The LibreView data management software is intended for use by both patients and healthcare professionals to assist people with diabetes and their healthcare professionals in the review, analysis and evaluation of historical glucose device data to support effective diabetes management. The LibreView software is not intended to provide treatment decisions or to be used as a substitute for professional healthcare advice.
§ The LibreLinkUp app is only compatible with certain mobile device and operating systems. Please check www.LibreLinkup.com for more information about device compatibility before using the app. Use of LibreLinkUp requires registration with LibreView. The LibreLinkUp mobile app is not intended to be a primary glucose monitor: home users must consult their primary device(s) and consult a healthcare professional before making any medical interpretation and therapy adjustments from the information provided by the app.
‡ 60-minute warm-up period required when applying the sensor.
₸ Sensor is water resistant in up to 1 metre (3 feet) of water for a maximum of 30 minutes. Do not immerse longer than 30 minutes. Not to be used above 10,000 feet.
The "Yes" link below will take you to a website other than Abbott Laboratories. Links to other websites are not under the control of Abbott Laboratories, and Abbott Laboratories is not responsible for the content of such websites or any link contained in such websites. Abbott Laboratories is providing these links to you only as a convenience, and the inclusion of any link does not imply endorsement by Abbott Laboratories of the site.
Do you want to leave this page?
If you're a healthcare professional located in a different country, please select your country's website from the list of our global sites or contact your local Abbott representative for correct information about the products available in your country.
If you're not a healthcare profressional, click here to go to the FreeStyle Libre consumer website for the UK.
Stay connected